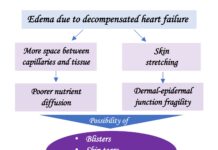
This week, two scientific meetings that I usually attend every year, have taken place: the 46th National Congress of Dermatology and Venereology in Palma and the EWMA 2018 Conference in Krakow.
It was a pleasure to chair a session about wound healing at the Spanish National Congress of Dermatology, where we talked about diagnostic and therapeutic pearls in the treatment of chronic leg ulcers.

This post is based on a pearl presented by my colleague and best friend Celia Horcajada: “The replacement of acenocoumarol by another oral anticoagulant in patients with Martorell ulcer”. Curiously, that same day, in Krakow, Jürg Hafner gave a talk in which this pearl had to be mentioned as well.

Martorell ulcer, as proposed by Dr. Jürg Hafner in his excellent article Calciphylaxis and Martorell Hypertensive Ischemic Leg Ulcer: Same Pattern – One Pathophysiology,1 is a hypertensive ischemic arteriosclerosis that can be framed within the clinical-physiological spectrum of calciphylaxis. In fact, both entities share, as we can see in the table, risk factors, clinical and histological aspects.
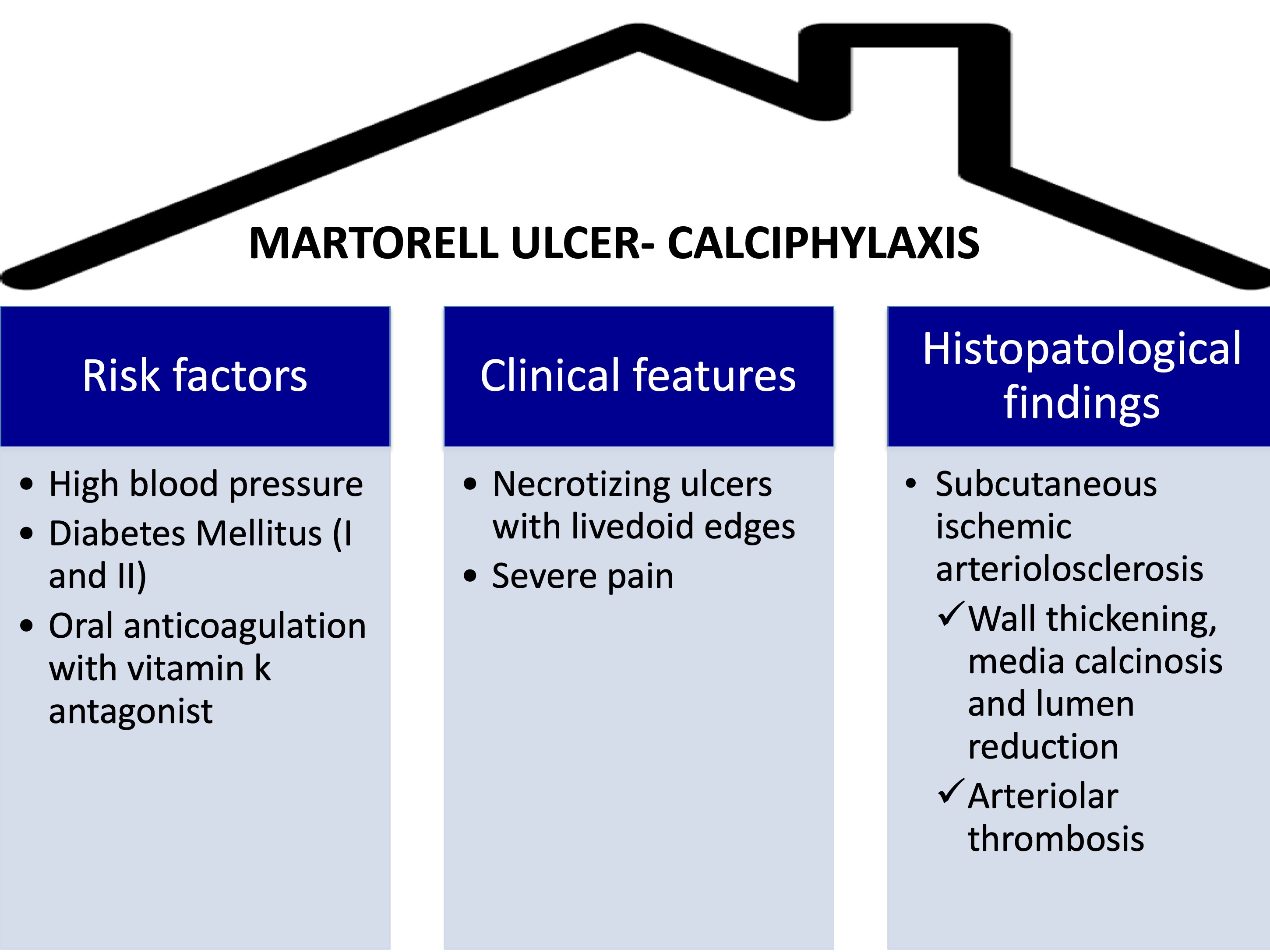
This concept is relevant to better understand the physiopathology of these two entities and to propose a therapeutic approach with common strategies, such as debridement, negative pressure therapy, early grafting or sodium thiosulfate. Furthermore, in both Martorell ulcer and calciphylaxis it is essential to control cardiovascular risk factors and assess the replacement of the vitamin K antagonist by low molecular weight heparin or factor Xa inhibitors.
And why is it interesting to withdraw acenocoumarol in these patients?
Acenocoumarol has an anticoagulant action because it inhibits the activation of coagulation proteins that depend on vitamin K (prothrombin, factor VII, IX, X). Because of this dependence on vitamin K, these coagulation factors are part of a family of proteins called G-proteins. This vitamin K-dependent protein group also includes anticoagulant factors, such as vitamin S or C (so in patients with a deficit of these factors, skin necrosis can occur due to acenocoumarol) and the calcification-inhibiting protein complex Fetuin A (or glycoprotein α2-Heremans-Schmid)-GLA-matrix protein (or MGP). In order for the binding and subsequent activation of this protein system to take place (as with all other Gla-proteins), the glutamic residues of the GLA-matrix protein have to undergo vitamin K-dependent carboxilation. For this carboxilation to be possible, the vitamin K has to be converted to its reduced form by an epoxide-reductase enzyme.
Acenocoumarol blocks the action of the epoxide-reductase, so it inhibits the activation of this transport protein complex responsible for binding insoluble calcium phosphate and inhibiting pathological calcification.
These proteins were discovered in the 1990s and their existence explained the nephrologists’ hypothesis that vitamin K antagonists could worsen cases of calciphylaxis. The way this calcification inhibitor complex acts is not completely clear, but its role seems key to prevent ectopic calcification in patients with predisposing pathologies. Vitamin K deficiency and the consequent decrease in the concentration of carboxylating GLA matrix protein have been associated with an increased risk of calciphylaxis.2
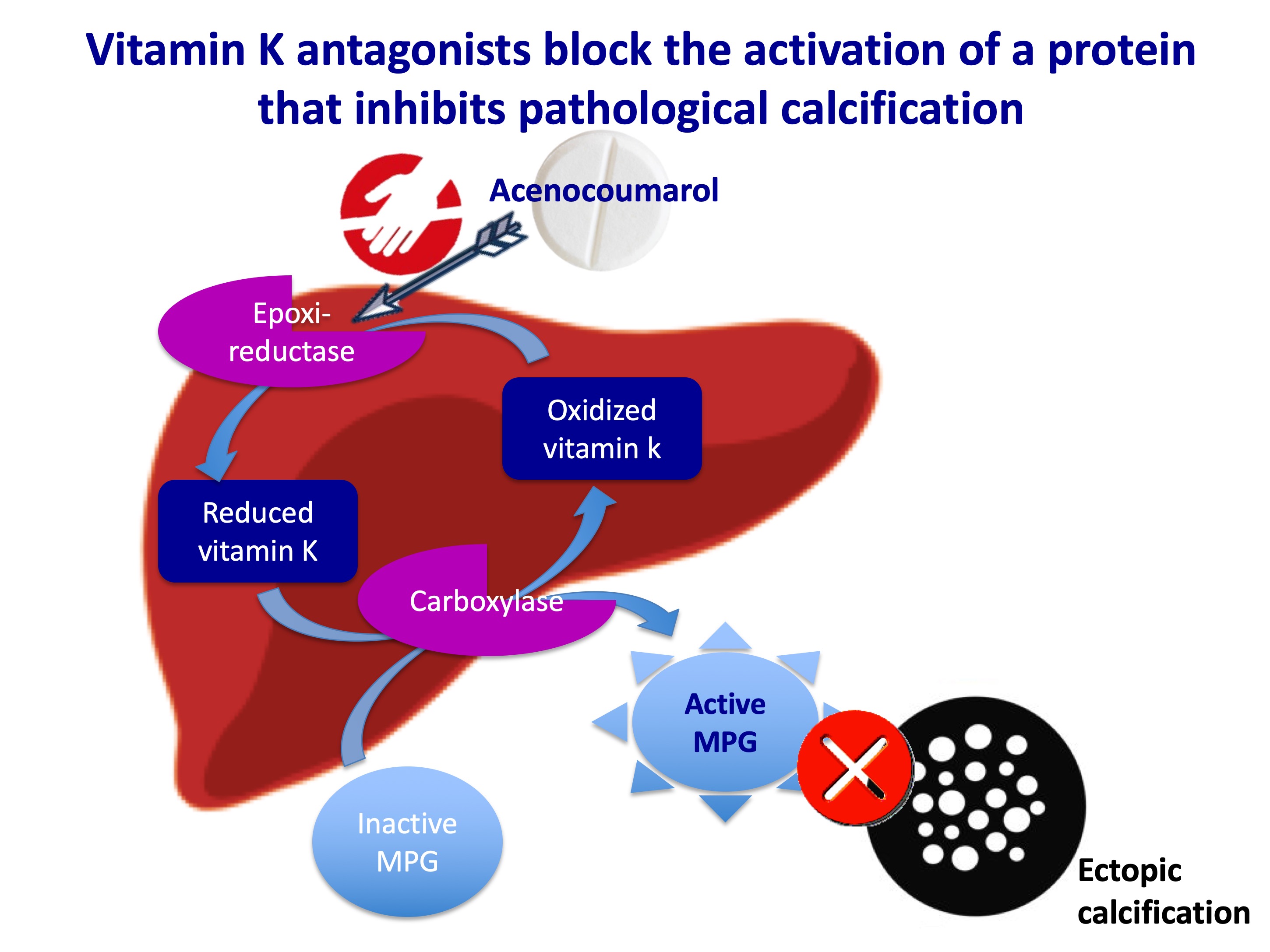
Therefore, in patients being treated with acenocoumarol in whom we suspect lesions in the ulcer spectrum of Martorell-calciphylaxis, replacement with another anticoagulant (low molecular weight heparin or activated factor X inhibitors) is recommended.
References:
- Hafner J. Calciphylaxis and Martorell Hypertensive Ischemic Leg Ulcer: Same Pattern – One Pathophysiology. Dermatology. 2016;232(5):523-533.
- Nigwekar SU, Bloch DB, Nazarian RM, Vermeer C, Booth SL, Xu D, Thadhani RI, Malhotra R. Vitamin K-Dependent Carboxylation of Matrix Gla Protein Influences the Risk of Calciphylaxis. J Am Soc Nephrol. 2017 Jun;28(6):1717-1722.
Also available in: Español (Spanish)